Do Supplements Work? Evidence-Based Guide to Health
Do supplements work? Discover the truth about supplements with clear evidence, dose/form guidance, and label checks. Spot proprietary blends, fake reviews, and detox myths; verify USP/NSF quality; learn when products help—or don’t. Make smarter, safer choices today.
“Natural” products promise health in a bottle. Do supplements work? From mood-boosting gummies to detoxifying powders? A well-lit before-and-after photo flashes across your feed, and suddenly your brain whispers, “This is the fix.” Our attraction to supplements isn’t accidental; it’s hard-wired by cognitive biases and reinforced by persuasive marketing. But the question remains: Do supplements work? See what’s worth your money
This article sorts fact from hype. We’ll dissect the psychological tricks and legal loopholes used to sell supplements, explain why “natural” means less than you might think, and show you how to recognise real evidence. You’ll also learn when supplements can truly help and how to choose them safely.
Why We Believe the Hype
Cognitive biases
Our brains look for shortcuts. Confirmation bias causes us to seek out information that supports our existing beliefs and overlook contradicting data. This is why a few positive reviews can feel more compelling than a solid clinical trial. The halo effect occurs when a product’s clean design, celebrity endorsement, or “clinically studied” wording leads us to assume it works—even if the formula is weak (NCCIH. Placebo Effect. January 2020). A third bias, naturalness bias, makes us equate “natural” with safer or better, even though the FDA does not evaluate supplements for safety or effectiveness before they hit the market. (Antonio J. Dietary supplements. PMC, 2025)
Bias breaker: Combat these biases by checking whether the product has been tested in randomized, blinded human studies. Start with reputable sources like the National Institutes of Health’s Office of Dietary Supplements. Then read the Supplement Facts panel and look for independent quality seals.
Bias breaker (quick, illustrative example — not a recommendation):
If you’re considering a Vitamin D3 from Nature Made, first pause and ask yourself: Do supplements work? — for my specific goal and labs? Then run this 60-second check:
- Supplement Facts: Confirm the active ingredient (cholecalciferol D3), the dose (e.g., 1,000 IU / 25 mcg), and the serving size.
- Independent quality: Look for a USP Verified mark on the label and verify it on USP’s directory (don’t rely on packaging alone).
- Human evidence: Scan the product page or brand materials for randomized, blinded human studies that align with your goal (e.g., correcting a documented vitamin D deficiency), not just marketing claims.
- Clinical fit & safety: Match the dose/form to guideline ranges, review warnings and drug interactions, and avoid stacking multiple products that duplicate vitamin D.
This is a brief educational example using Nature Made to illustrate due diligence. It is not an endorsement or a purchase recommendation. Always consult your healthcare provider before starting any supplement.
Social proof and influencer marketing
When we’re uncertain, we copy people we trust. Likes, shares, and glowing testimonials give the impression that a supplement must work. Research shows that relatable influencers can increase purchase intent, yet this doesn’t answer whether the product is effective. In the U.S., the Federal Trade Commission requires influencers to disclose material connections with brands. Posts must be clearly labelled as ads or paid partnerships; hidden disclosures are a red flag(Federal Trade Commission. Fake reviews and testimonials rule. 2024). Always ask: Is this genuine advice or a sponsored post?
The lure of quick fixes
Humans favour immediate rewards over delayed benefits. Marketers exploit present bias by promising “30‑day transformations” or “detox cleanses.” But for major outcomes like preventing heart disease or cancer, top U.S. guidelines find little benefit from most vitamins and minerals(ACC. USPSTF on beta-carotene, vitamin E for CVD and cancer. 2022). Pause when you see a miraculous claim and look up the evidence before you buy.

The Allure of “Natural”: Decoding Labels
Natural vs. organic vs. plant‑based vs. clean label
Natural: The FDA hasn’t issued a formal definition for “natural,” but has a long‑standing policy: the term may be used if the food contains no added artificial or synthetic ingredients (FDA. Natural on food labeling. 2018). This says nothing about farming methods, safety, or health benefits. For supplements, companies may use “natural” as marketing; they are responsible for safety and labeling but do not need FDA approval before sale(PMC. Dietary supplements misconceptions. 2025).
Organic: USDA organic certification regulates how ingredients are produced. “100 percent organic” products must be entirely organic, “organic” products must contain at least 95 percent organic ingredients, and “made with organic” products contain at least 70 percent(USDA. Organic labeling). Organic tells you about farming practices (no genetic engineering, ionizing radiation, or sewage sludge)(USDA. Organic labeling), not whether a supplement works.
Plant‑based: Plant‑based products are made from plant ingredients. FDA guidance on plant‑based milk alternatives notes that composition and nutrient profiles vary by source(FDA. Plant-based milk and food alternatives. 2025); the term does not guarantee nutritional equivalence or purity. Similarly, a “plant‑based” supplement could be underdosed or contaminated.
Clean label: There is no legal or regulatory definition for “clean label.” Consumers often read it as short, simple ingredient lists with no artificial additives and more transparency, yet expert reviews show the term remains ambiguous and poorly understood by many shoppers. Treat “clean” as marketing; you still need to verify dose, evidence, and third-party testing. (UGA Cooperative Extension, IFT)
Does “natural” mean safer?
No. Many plants are toxic. NCCIH highlights that some “natural” products—such as kava or ephedra—are linked to severe liver injury and cardiovascular events(NCCIH. Natural vs synthetic drugs). The FDA banned ephedra‑containing supplements after reports of deaths. Likewise, natural supplements can interact with medications or cause side effects. Always consult a healthcare provider.
Capsule shells: animal vs. plant
Supplement hard capsules are most commonly made from gelatin (animal-derived collagen) or hypromellose/HPMC (a cellulose-derived, vegan-friendly material). You may also see pullulan (a fermentation-derived vegan option). Softgels are typically gelatin, though vegetarian versions use modified starch blends (e.g., starch + carrageenan + glycerin). “Plant-based” does not automatically mean acid-resistant—if you need protection from stomach acid, look for “enteric” or “delayed-release” on the label. Always check the “Other ingredients” for colorants/opacifiers (e.g., titanium dioxide), coatings (e.g., shellac), and for vegan/vegetarian, halal, or kosher certifications. (This clarifies that HPMC is cellulose-derived and that pullulan/modified starch systems are common vegetarian alternatives.)
Placebo and Nocebo Effects
Placebo effect: When you expect a supplement to help, your brain and body may generate real improvements in symptoms. Rigorous trials use blinding to distinguish chemical effects from expectation effects. The placebo effect is driven by context—caregiver attention, packaging and positive framing(NCCIH. Placebo effect. 2020).
Nocebo effect: The opposite happens when you anticipate side effects; you may experience adverse symptoms even when taking an inert substance(PMC. Nocebo effect. 2025) Media reports or online chatter about “detox” reactions can seed negative expectations.
Practical tip: If you’re testing a new supplement, keep a symptom diary. Track one primary outcome for a set period (e.g., two weeks), maintain your usual routines, and if possible try an inert look‑alike on alternate weeks. This can help differentiate real effects from expectations and natural fluctuations.
Placebo and Nocebo Effects
Placebo effect. When you expect a supplement to help, your brain–body systems can produce real, measurable improvements (less pain, calmer mood, better sleep). That’s why good studies use randomization and blinding—to separate the pharmacologic effect of an ingredient from the expectancy effect created by context (the clinician’s attention, confident language, polished packaging, and positive framing).
Nocebo effect. The flip side is that expecting side effects can trigger symptoms even with an inert substance. Common nocebo-driven complaints include headache, nausea, bloating, and fatigue. Sensational media posts or “detox horror stories” prime negative expectations and make nocebo more likely.
Why this matters for supplements. Improvements (or new symptoms) after starting a product aren’t proof—by themselves—that the ingredient works (or harms). Without blinded, randomized human trials, it’s hard to know whether you’re seeing drug action or expectations.
A quick at-home check (simple, plain-English)
Goal: tell if changes you feel are from the supplement or just expectations/normal ups-and-downs.
Important: this is educational only and not medical advice. Stop if you feel unwell and talk to your clinician.
Option 1 — Easiest (no blinding)
- Pick ONE thing to measure.
Examples: minutes to fall asleep, pain score (0–10), number of bowel movements/day, workout reps. - Baseline (7 days): don’t take the supplement. Record the number each day.
- Week A (7 days): take the supplement exactly the same time each day. Keep sleep, caffeine, meals, and training the same. Record daily.
- Compare averages:
- If Week A’s average is consistently better than Baseline (ideally ≥10–20% and on most days, not just one day), the supplement might be helping.
- If there’s little/no difference, it’s likely expectations or normal fluctuation.
Option 2 — Better (mini A/B test)
- Do Week A (supplement) then Week B (no supplement), or the reverse.
- Keep routines identical.
- Compare Week A vs Week B averages. A clear, repeatable edge for the supplement week suggests a real effect.
Want to get fancy? If you can make look-alike capsules (one real, one empty) and have a helper label them “A/B” so you don’t know which is which, that’s a blinded version and reduces placebo/nocebo.
Quick example (sleep onset)
- Metric: minutes to fall asleep.
- Baseline avg (7 days): 45 min
- Week A (supplement) avg: 31 min
- Change: 45 → 31 = 14 min faster (~31%), and 5/7 days were better than baseline → likely meaningful
- If Week B (no supplement) avg: 44 min → effect looks repeatable (A better than both Baseline and B)
How to reduce nocebo while you test
- Use neutral language with yourself (avoid “this will make me sick”).
- Don’t doom-scroll scary “detox reaction” posts during the test.
- Keep one change at a time (no new diet/workout/coffee shifts).
How Misinformation Spreads
“Clinically studied” claims without context
Many brands tout that their ingredients are “clinically studied” or “clinically proven.” Yet they rarely specify the study design, sample size or results. Competent evidence usually means randomized, blinded human trials with pre‑specified outcomes. Be wary of claims based on one small study, animal research or surrogate markers. Look for human trials with absolute risk reductions rather than relative percentages to understand real‑world benefit.
Cherry‑picking and spin
Selective reporting hides null results and highlights favourable subgroups. Marketers might show only the best graph or report a relative 50 percent improvement without revealing that the absolute difference is tiny. Always ask: What was the baseline? How many people improved?
Algorithms and fake reviews
Social‑media feeds show more of what you interact with, creating echo chambers. Watch a supplement video once, and you’ll see it everywhere. The FTC’s 2024 final rule prohibits businesses from selling fake or manipulated reviews, buying positive or negative reviews, or using undisclosed insider testimonials (FTC. Fake reviews and testimonials rule. 2024). It also bans fake social‑media indicators and company‑controlled review sites. If reviews feel too uniform or appear soon after a product launch, be skeptical.
Sponsored content and disclosure
In the U.S., influencers must clearly label paid posts with terms like “Ad” or “Paid partnership.” Hidden hashtags or buried disclosures violate FTC guidelines. If you can’t tell whether a post is sponsored, treat it as marketing and look elsewhere for evidence.
Red Flags to Avoid
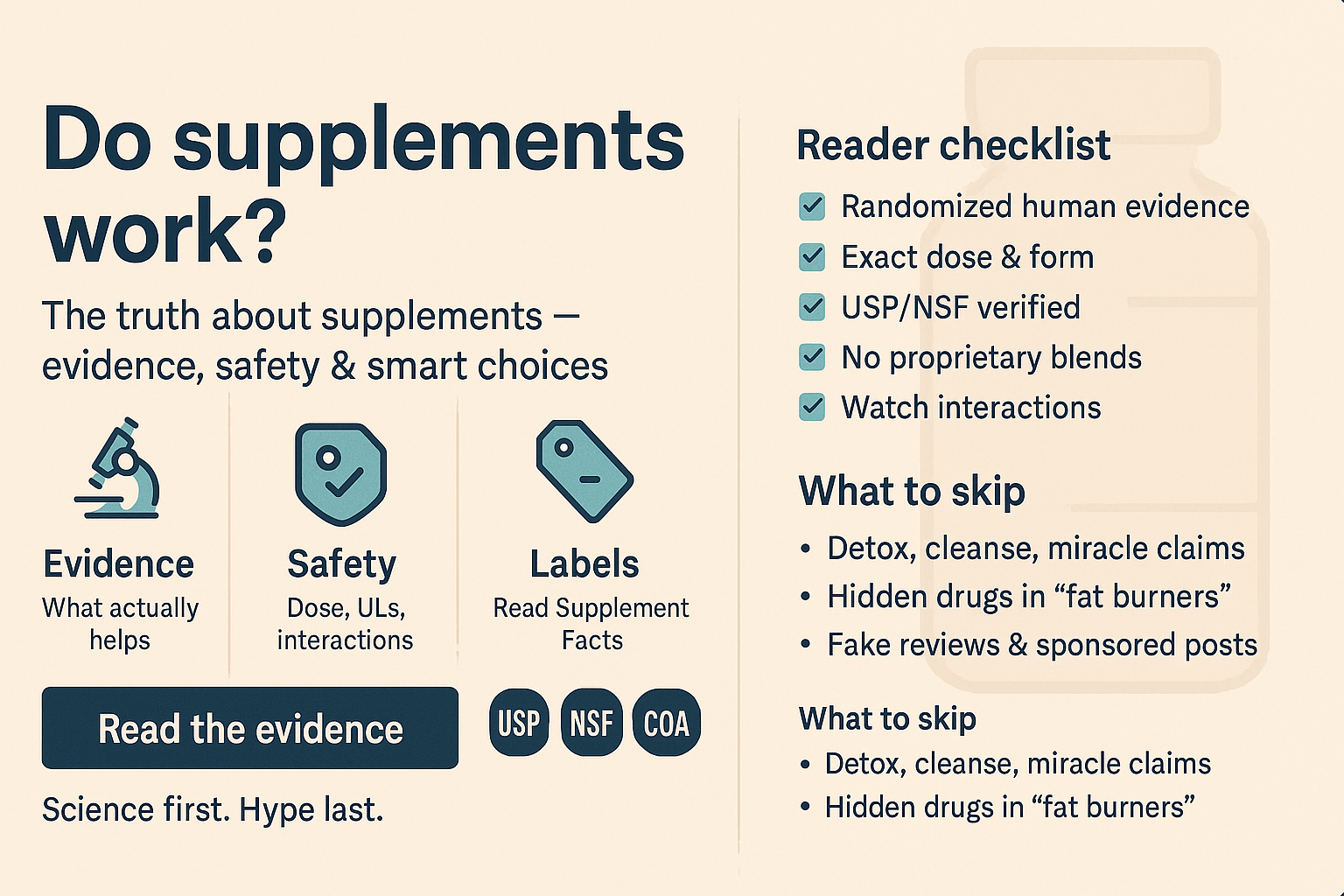
Too‑good‑to‑be‑true claims: Any product promising rapid weight loss, muscle gain or “detoxification” without effort is likely marketing. The FDA has found hidden drugs in so‑called “fat burners” and “male enhancement” supplements(PMC. Dietary supplements misconceptions. 2025).
Detox promises: Your liver and kidneys detoxify your body. No supplement can magically cleanse “toxins” from a healthy body. Claims of flushing heavy metals or resetting metabolism are unproven.
Proprietary blends: These list ingredients without individual doses, so you can’t verify whether each component is present at an effective level. Manufacturers use them to hide low amounts.
Miracle before/after photos: Lighting, posing and editing can exaggerate results. Without randomized trials, images are just ads.
Missing third‑party testing: Look for the USP Verified or NSF Certified for Sport seal. USP audits facilities, reviews documentation and tests products for identity, potency and purity(Pharmacy Times. USP verified mark. 2014). NSF Certified for Sport screens supplements for 290+ banned substances and confirms that contents match the label (NSF Sport. What our mark means).
Red Flags to Avoid: Do supplements work?
1) Too-good-to-be-true claims
Any product promising rapid fat loss, effortless muscle gain, or a “30-day body reset” is marketing, not science. The FDA has repeatedly found hidden drugs in “fat burners” and “male enhancement” supplements [16].
Do instead: Ignore hype timelines; look for human data (randomized, blinded trials), realistic effect sizes, and clear dosing.
2) “Detox” promises
Your liver and kidneys already detox you. No over-the-counter supplement can “flush toxins,” “reset metabolism,” or “pull heavy metals” in healthy people. Many “detox” blends are just laxatives/diuretics, which shift water weight—not toxins. Medical chelation (for true heavy-metal poisoning) is doctor-supervised only.
Do instead: Save your money; prioritize diet, hydration, sleep, and clinically indicated care.
3) Proprietary blends (no amounts per ingredient)
A “proprietary blend” lists a total mg for multiple ingredients without showing each ingredient’s dose. You can’t verify if any component meets effective levels; under-dosing is common.
Spot it: “Proprietary blend 900 mg (X + Y + Z…)” with no per-ingredient mg.
Do instead: Choose labels that disclose exact mg per ingredient. If a brand refuses to share amounts or a recent third-party COA (certificate of analysis) tied to your lot number, skip it.
4) Miracle before/after photos
Lighting, posing, diuretics, and editing can fake dramatic changes. Pictures aren’t data.
Do instead: Ask for study design, sample size, endpoints, and absolute numbers, not just relative percentages.
5) Missing independent testing
“Lab tested” or a glossy “COA” jpeg isn’t enough. Look for recognized programs and verify them:
- USP Verified — audits facility quality systems; tests identity, potency, purity [15].
- NSF Certified for Sport — verifies label claims and screens 290+ banned substances [14].
Do instead: Check the USP/NSF directories; confirm your exact product/lot appears there.
Extra red flags savvy readers ask about
- “FDA approved” supplement → Red flag. In the U.S., dietary supplements are not FDA-approved for safety or effectiveness before sale.
- “Clinically proven” with an asterisk* → Often refers to a study on one ingredient (or in animals/small samples), not the finished product.
- Mega-doses beyond the Tolerable Upper Intake Level (UL) → Higher dose ≠ better; increases side-effect risk.
- “Medical grade” → Not a regulated term for supplements; treat as marketing.
- Vague extracts (“herbal complex,” “standardized extract”) without marker compounds or % standardization → You can’t judge quality or equivalence.
- GMP logos with no audit body → Prefer audited GMP (e.g., NSF/UL/NPA) over self-claims.
- QR code to “lab report” that lacks lab name, method, date, lot number, or pass/fail specs → Not reliable.
30-second Label Audit (keep this handy)
- Supplement Facts present? Ingredient names + mg per serving (not just totals).
- Dose & form match evidence? (e.g., magnesium glycinate at a studied mg, not “blend”).
- Independent seal? USP Verified / NSF Certified for Sport and verifiable in the directory.
- No disease claims. Structure/function claims are OK (“supports…”); “treats/cures” = red flag.
- Other ingredients: unnecessary colorants, shellac, or allergens; capsule type (gelatin vs HPMC/pullulan) per your dietary needs.
Mini-FAQ (fast answers readers usually want)
What does “proprietary blend” mean?
A proprietary blend lists multiple ingredients under one total weight without the exact per-ingredient amounts, making it impossible to verify effective doses and often masking under-dosing.
What about proprietary blends to “protect IP”?
“Protecting IP” is a common claim, but quality brands still disclose per-ingredient milligrams or share a lot-specific COA on request. If they won’t, choose a more transparent product.
Does “no USP/NSF seal” mean the product is bad?
Not automatically—but risk is higher. Prefer brands that use recognized, independent testing.
Are gummies effective?
Sometimes, but they’re often underdosed and less stable. Check serving size (often 2–4 gummies), sugar load, and per-ingredient mg.
Is “natural” safer?
Not necessarily. “Natural” ≠ safe or effective. Some botanicals have serious interactions; always review warnings/drug interactions.
Bottom line: If a product makes big promises, hides exact doses, lacks independent verification, or leans on photos and buzzwords instead of data—walk away. Use clear labels, human evidence, and third-party testing to guide your choice
When Supplements Work: Evidence-Based Uses
Correcting deficiencies (best-supported)
- Folic acid (vitamin B9). Taking 400 μg daily before conception and in early pregnancy prevents neural-tube defects—one of the strongest, clearest uses of a supplement (CDC. Folic acid. 2025)Some people prefer methylfolate, but folic acid remains the standard recommendation.
- Iron. Treats iron-deficiency anemia; the form and dose depend on labs and symptoms. Because excess iron can be harmful, don’t start without confirmed deficiency and clinician guidance.
- Vitamin B12. Oral B12 works as well as injections for normalizing levels and is usually easier and cheaper—so for B12 deficiency, the short answer to Do B12 supplements work is yes when the deficiency is confirmed and dosing is appropriate. (PMC. Oral vs intramuscular vitamin B12. 2018)
- Deficiency is more common in vegans, older adults, and malabsorption—use blood tests to set dose.
Performance and specific conditions
- Creatine monohydrate. Consistently boosts phosphocreatine stores, improving max strength, sprint performance, and lean mass with minimal side effects (mainly water weight) (PMC. Creatine for exercise and performance. 2021). Typical dose: 3–5 g/day; useful for vegetarians/vegans and high-intensity athletes.
- Melatonin.Low doses (0.5–5 mg) at local bedtime help reduce jet-lag symptoms after east–west travel (NCCIH. Melatonin: what you need to know. 2024) For chronic insomnia, the evidence is mixed—get sleep-specialist input before long-term use.
- Probiotics. Certain strains—e.g., Lactobacillus rhamnosus GG and Saccharomyces boulardii—lower the risk of antibiotic-associated diarrhea by ~40% when taken at studied doses; benefits are strain- and condition-specific(PMC. Probiotics and antibiotic-associated diarrhea. 2025)
- Prescription EPA (icosapent ethyl). A purified eicosapentaenoic acid drug that lowers triglycerides and, at 4 g/day, reduces cardiovascular events in high-risk patients on statins(PMC. Icosapent ethyl in cardiovascular risk reduction. 2023). This is prescription-only, not an over-the-counter fish-oil capsule.
Who may not benefit (or may be harmed)
- General multivitamins/antioxidants. In well-nourished adults, large trials show no prevention of heart disease or cancer; the USPSTF recommends against beta-carotene and vitamin E for primary prevention (beta-carotene may raise lung-cancer risk in smokers; vitamin E shows no benefit) (ACC. USPSTF on beta-carotene & vitamin E. 2022)
- “Detox,” “cleanse,” and “fat-burner” formulas. Often contain stimulants or undisclosed ingredients; the FDA regularly issues warnings and recalls for safety concerns (PMC. Dietary supplements misconceptions. 2025).
How to Choose Supplements Safely: A Step‑by‑Step Checklist
Identify your goal and check evidence. Look for randomized controlled trials or systematic reviews on reputable databases (PubMed, Cochrane, NCCIH). Avoid basing decisions on testimonials.
Match the dose and form. Verify that the supplement provides the same ingredient, dosage and chemical form used in human studies. Beware of proprietary blends.
Read the Supplement Facts panel. Check serving size, number of capsules per dose, and any added ingredients. For vegans or people with dietary restrictions, confirm whether the capsule is gelatin or plant‑based(PMC. Vegetarian vs nonvegetarian capsules. 2017).
Look for independent quality seals. Products bearing the USP Verified or NSF Certified for Sport mark have undergone third‑party testing for identity, potency and purity. (Pharmacy Times. USP verified mark. 2014) (NSF Sport. What our mark means)Use the USP or NSF online directories to verify the certificate.
Check regulatory disclosures. In the U.S., labels must include a disclaimer: “This product is not intended to diagnose, treat, cure or prevent any disease.” Claims beyond structure/function are illegal. Disregard products making drug‑like claims (e.g., “cures depression”).
Assess interactions and safety. Consult a healthcare provider or pharmacist, especially if you take prescription medications, are pregnant or breastfeeding, or have chronic conditions. Some supplements can alter drug metabolism or blood clotting.
Monitor your response. If you decide to try a supplement, run a personal trial lasting a few weeks, track a single outcome, and stop if no benefit appears or if side effects occur. Remember placebo and nocebo effects.
Beware of marketing traps. Don’t be swayed by celebrity endorsements, “all‑natural” labels or flashy packaging. Facts, not feelings, should drive your decision.
Recommendations: Supplements Worth Considering (Educational, Not Prescriptive)
If you decide to explore supplements, focus on those with strong evidence and clear indications. Examples include:
Prenatal folic acid: People who might become pregnant should take 400 µg of folic acid daily to prevent neural‑tube defects[7]. Prenatal formulas often contain this dose, but check labels and avoid proprietary blends. Those with a history of neural‑tube defects may require higher doses under medical supervision.
Iron and vitamin B12 for diagnosed deficiencies: If lab tests show iron‑deficiency anaemia or vitamin B12 deficiency, supplementation is effective. B12 can be taken orally or via injection(PMC. Oral vs intramuscular vitamin B12. 2018). Choose forms like ferrous bisglycinate or methylcobalamin that match research protocols. Always work with a clinician for dosing and follow‑up testing.
Creatine monohydrate for athletic performance: Do creatine supplements work? Athletes and older adults seeking to improve high-intensity strength or muscle mass may benefit from 3–5 g/day of creatine monohydrate (PMC. Creatine for exercise and performance. 2021) . Look for plain creatine monohydrate powder with no proprietary blends, ideally USP Verified or NSF Certified for Sport.
Evidence‑based probiotics: For preventing antibiotic‑associated diarrhoea, choose strains supported by research—such as Lactobacillus rhamnosus GG or Saccharomyces boulardii—at doses used in studies(PMC. Probiotics and antibiotic-associated diarrhea. 2025). Products should list the strain and colony‑forming units and carry a quality seal.
Melatonin for jet lag: If you travel across time zones, low‑dose melatonin taken at your destination’s bedtime may reduce jet‑lag severity(NCCIH. Melatonin: what you need to know. 2024). Avoid using high doses or long‑term melatonin without professional guidance.
These recommendations illustrate how to match specific supplements to specific needs. They are not a blanket endorsement; always consult a healthcare provider.
Conclusion
Supplements occupy a grey zone between food and medicine. The industry thrives on cognitive biases, persuasive marketing and regulatory gaps. Yet some supplements deliver real benefits when chosen wisely. To uncover the truth about supplements:
Slow down.Check human evidence and read labels carefully. Be skeptical.Question marketing claims and look for third‑party quality marks. Consult professionals.Use supplements to address verified deficiencies or specific goals, not as panaceas. By applying these principles, you can cut through the noise and make decisions based on facts rather than hype.
Practical Takeaways and Safe Usage Notes
Verify need: Get lab tests or professional evaluations before starting supplements.
Check evidence: Use trusted databases; search for randomized, human trials.
Match dose and form: Ensure the product provides the studied ingredient at the studied dose.
Look for USP/NSF seals: Verify claims through public directories.
Watch for interactions: Consult healthcare providers, especially if pregnant, nursing, taking medications or managing chronic conditions.
Track your experience: Use a symptom diary to differentiate real effects from placebo/nocebo. Stop if adverse effects occur.
Storage matters: Store supplements according to label instructions and check expiration dates.
Regulation is limited: Understand that supplements are regulated post‑market; safety and efficacy are not reviewed before sale (PMC. Dietary supplements misconceptions. 2025).
Disclaimer
This article provides general information and does not replace professional medical advice. Always consult a qualified healthcare provider before starting or stopping any supplement, especially if you have underlying health conditions or take medications.
How We Checked the Evidence
We reviewed recent guidelines, systematic reviews and clinical trials from reputable sources. We relied on U.S. and international agencies (FDA, USDA, CDC, FTC), public health organisations (NCCIH, Office of Dietary Supplements), peer‑reviewed journals (e.g., Am J Cardiovasc Drugs, Nutrients), and authoritative reviews (Cochrane). Our sources were chosen for transparency, methodological rigor and recency. When evidence was conflicting or limited, we indicated uncertainty or noted that data were insufficient. All claims are accompanied by citations to primary sources or high‑quality summaries. No funding was received for this article, and we have no financial ties to supplement manufacturers.
How we reviewed this article
Sources
Our experts continually monitor the health and wellness space, and we update articles when new information becomes available.
Current Version
Aug 22, 2025
Written By
Nour Hany
Edited By
KirolosReda
Medically Reviewed By
DR. Rania Elserafy
Leave a comment
Your email address will not be published. Required fields are marked *

![Why Am I In A Calorie Deficit And Gaining Weight? [Fix]](https://wellness.culinarycreationss.com/storage/health-and-nutrition-1/why-am-i-in-a-calorie-deficit-and-gaining-weight-fix/4a48e795-580b-4097-aae8-42ae368bde7a.webp)
